- Blog
- Xee download mac
- 808 bass arturia analog lab
- Mustang pfd rearm kit
- Instagram for pc free download windows xp
- Hp envy intel centrino wireless n 2230 driver
- Hasi female version video song
- Hp psc 1350 driver download
- Endnote x7 compatibility
- Max payne 3 pc crwsh after brightness check
- Ifixit mac air 2011 battery
- Where do i find ps3 mac address
- Tony hawk pro skater 5 levels
- Convert flac to wav free no limitations
As a result, with the occurrence of freezing fronts, a typical freeze concentration profile with the peak concentration at the center bottom of a frozen bulk is unavoidable ( Maity et al., 2009 Kolhe and Badkar, 2011 Roessl et al., 2014 Reinsch et al., 2015).įreeze concentration was shown to be dependent on the freezing process rather than storage temperature ( Hauptmann et al., 2019). Additionally, this freeze concentration effect leads to natural convection due to density gradients ( Butler, 2002) and therefore settlement of solutes. While freezing, solutes are concentrated in front of the phase boundaries and partition between solid and liquid phase ( Bhatnagar et al., 2007), leading to macroscopic freeze concentration. During freezing of larger bulk volumes, ice fronts progress from cooled container walls toward the center of the container.

In large scale freezing operations, macroscopic freeze concentration leads to non-homogeneous solute distribution profiles. At a microscopic scale, crystallization of water molecules leads to freeze concentration of the remaining solutes within the ice crystal structure causing freezing temperature dependent phase behavior of proteins ( Wöll et al.,2019a,b). Protein activity loss or aggregation was correlated with freeze concentration ( Reinsch et al., 2015 Roessl et al., 2015). While freezing slows down and reduces degradation reactions of the API, freezing processes expose the protein to different stresses such as cold denaturation ( Privalov, 1990), freeze concentration ( Bhatnagar et al., 2007), ice crystal formation ( Chang et al., 1996), and potential excipient crystallization. Therefore, many biopharmaceuticals are stored in a frozen state ( Singh et al., 2009 Authelin et al., 2020). Due to their high value and production cost, activity loss of the active pharmaceutical ingredient (API) during shipment and storage has to be limited by selection of suitable formulation agents ( Chang et al., 2005 Bauer et al., 2017) and storage conditions.

Therapeutic proteins are among the top selling pharmaceuticals. Using a 2D-resolved temperature profile, a shift of the last point to freeze position from top to bottom of a container was observed when freezing above −30☌. As a hypothesis, we conclude that freezing temperature, from a freeze concentration view, is less important in containers with small characteristic freezing distances such as freeze bags. Furthermore, freezing time estimations based on the Plank equation revealed model limitations due to start-up temperature gradients, that can be corrected by an empirically extended Plank model. Freezing times varied from 35 to 81 min for temperatures between −60 and −20☌ and impacted freeze concentration profiles. The applicability of the method is demonstrated by process characterization of a novel small-scale freeze-thaw device with minimized boundary effects using freezing times of purified water and concentrated formulations. A method to detect freezing based on the second derivative of temperature measurements from Fiber-Bragg-Grating sensors is presented to overcome this issue. Currently, process characterization often relies on one or very few temperature probes that detect freezing times based on raw temperature, which is largely influenced by freezing-point depression in case of concentrated solutions. Process characterization of freeze-thaw operations at different scales should be conducted with attention to freezing time and boundary effects to ensure the product stability throughout the process and process development.
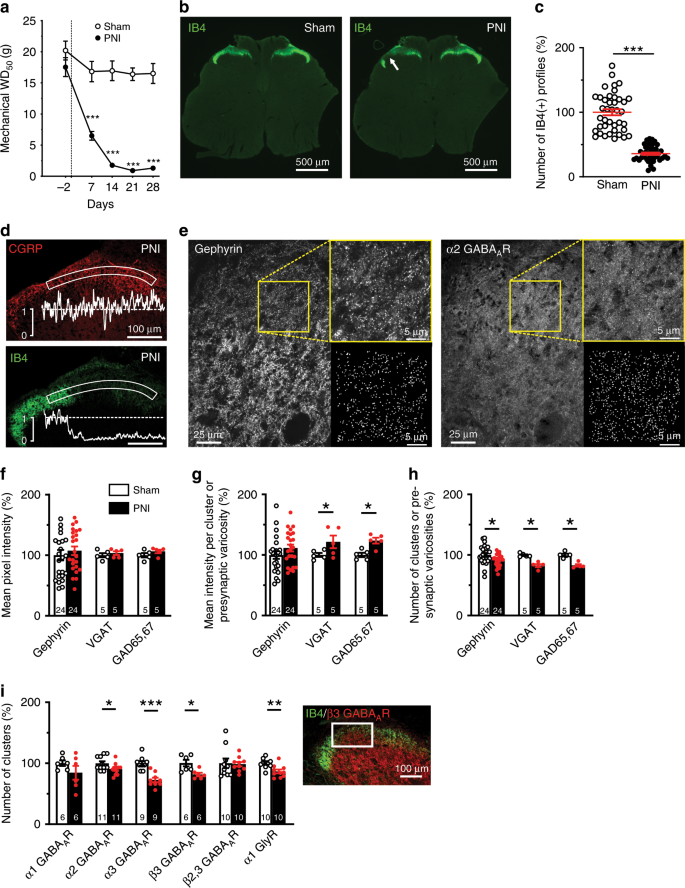
In biopharmaceutical production processes, freeze-thaw operations are used to ensure product integrity during long hold times, but they also introduce additional stresses such as freeze concentration gradients that might lead to a loss of protein activity.
- Blog
- Xee download mac
- 808 bass arturia analog lab
- Mustang pfd rearm kit
- Instagram for pc free download windows xp
- Hp envy intel centrino wireless n 2230 driver
- Hasi female version video song
- Hp psc 1350 driver download
- Endnote x7 compatibility
- Max payne 3 pc crwsh after brightness check
- Ifixit mac air 2011 battery
- Where do i find ps3 mac address
- Tony hawk pro skater 5 levels
- Convert flac to wav free no limitations
